|
Influence de la nature des électrodes sur la production d'oxyde d'azote dans l'arc électrique. Recherches sur l'action chimique des décharges électriques I. Le rendement réel de la production de l'ozone par l'effluve et son amélioration. size followed, according to Thomson, from Lenards results concerning their mean free path outside the cathode ray tube. Recherches sur l'action chimique des décharges electriques III. ACTION OF HIGH SPEED ELECTRONS ON METHANE, OXYGEN AND CARBON MONOXIDE. THE ACTION OF HIGH-SPEED CATHODE RAYS ON THE SIMPLER ALCOHOLS, ALDEHYDES AND KETONES, AND ON ETHYLENE. First, experiments by Lenard and by himself. If you want to repeat the experiment take the precautions necessary (very high Voltage, use gloves, etc) For the experiment we have to extract the electron beam which usually is directed toward the fluorescent screen of a cathode tube.In a TV or PC monitor tube the electrons flow in the direction indicated in fig. Ionizing Radiation Applied to Chemical Processes and to Food and Drug Processing. His corpuscle hypothesis seems based on only two experimental results. Ozone hazards incurred by electron Van de Graaff operation. The International Journal of Applied Radiation and Isotopes 1964, 15 The explanation is of course very simple - each electron in the cathode rays gains a kinetic energy of eV on accelerating down the tube before crashing into the. The formation of ozone in the radiolysis of gaseous oxygen. This hydrometallurgical process may promise a candidate for the effective separation and recovery of metal values from the sulfuric acid leaching liquor. 
Recovery efficiencies could be attained as follows: 98.7% for Ni 97.1% for Mn, 98.2% for Co and 81.0% for Li under optimized experimental conditions. So, the cathode ray experiment is also commonly known as J.J. The above modified experiments were performed by J.J. This also helped scientists in finding the charge of electrons. This ushered in a model of atomic structure referred to as the plum pudding model. Cathode rays also get deflected from their path if a magnetic field is applied. Dalton thought that atoms were indivisible particles, and Thomson's discovery of the electron proved the existence of subatomic particles. = using ammonium oxalate solution and saturated sodium carbonate solution, respectively. Explanation: Thomson's experiments with cathode ray tubes helped him to discover the electron (which Dalton did not know about). Moreover, the possibility to treat simultaneously both CRT and lamp powders is very important and interesting from an industrial point of view since it could be possible to run a single plant treating fluorescent powder coming from two different electronic wastes. In any case, the purity of final products from CRT, lamps, and MIX is greater than 95%. Yttrium loss can be reduced to minimum changing the experimental conditions with respect to the case of the more » CRT process. The lower yields obtained during treatments of MIX and lamp powders are probably due to the co-precipitation of yttrium together with other metals contained in the lamps powder only. 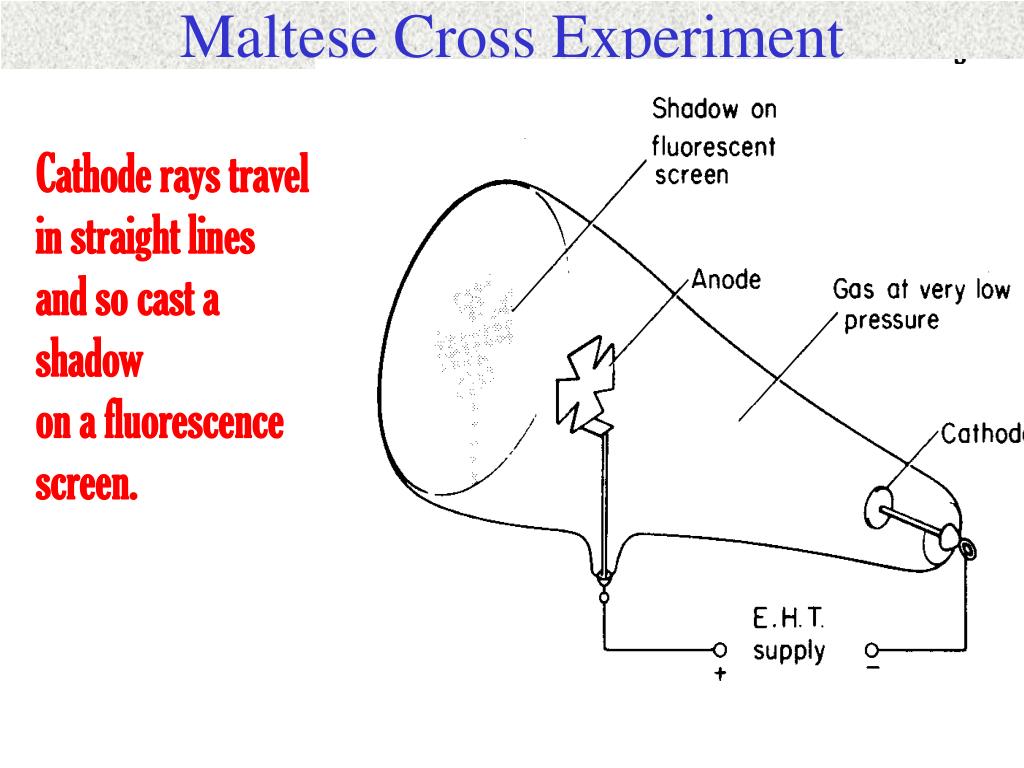
In the optimal experimental conditions, the recoveries of yttrium oxide are about 95%, 55%, and 65% for CRT, lamps, and CRT/lamp mixture (called MIX) powders, respectively. Experimental results have shown that process conditions necessary to purify the solutions and recover yttrium strongly depend on composition of the leach liquor, in other words, whether the powder comes from treatment of CRTs or lamp. The process for treating these materials includes the following: (a) acid leaching, (b) purification of the leach liquors using sodium hydroxide and sodium sulfide, (c) precipitation of yttrium using oxalic acid, and (d) calcinations of oxalates for production of yttrium oxides. Abstract: In this paper, yttrium recovery from fluorescent powder of lamps and cathode ray tubes (CRTs) is described. 
All different metals give off cathode rays. The particles that make up cathode rays are a 1000 times smaller than hydrogen atom. The cathode ray is composed of negatively charged particles. Economic simulation for the processes to recover yttrium from WEEE. Thomson made the following CONCLUSIONS from his experiment : 1.Recovery of yttrium from fluorescent powders. Electrostatic effect cathode ray tube : Demonstrates the phenomenon that the cathode ray deflects in the electric field, which proves that the cathode ray is a. 
The issues involved can be so intricate, in fact, that a leading expert in Hertzs experimental practice, Jed Buchwald (1995), appears to have misjudged the case of attempts to replicate Hertzs cathode ray experiments. Fluorescent powder of cathode ray rubes. IntroductionAdjudicating questions of experimental replication is an extremely difficult and subtle matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
